Jan 5, 2025
THEORETICAL FRAMEWORK:
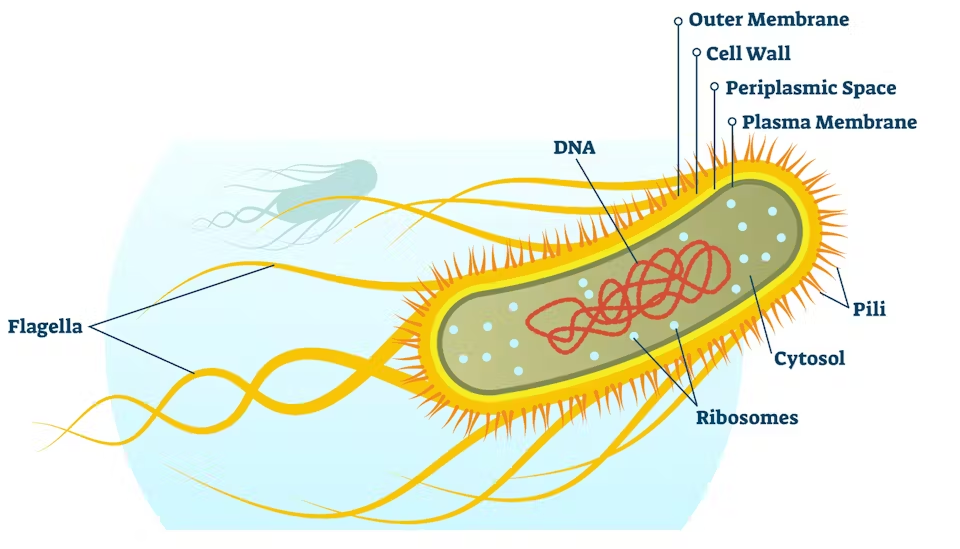
1 E. coli Bacteria
The bacteria which was used was developed by scientist Douglas Hanahn to maximize the effect of genetic transformation. This was with the alteration of the Rec A1 genes. This disabled enzyme recombination and the inactivation of homologous recombination, and End A1, to inactivate the intracellular endonuclease, both to prevent plasmid degradation.
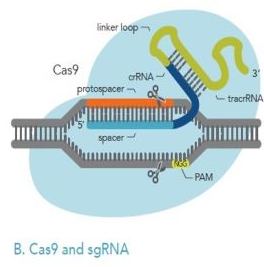
2 gRNA
The gRNA is a guide RNA composed of crRNA and tracrRNA, which can be single, sgRNA, or not be a gRNA. The crRNA is a sequence of 17 to 20 nucleotides complementary to the target DNA. The tracrRNA is the binding structure for the Cas9 nuclease. This combination utilizes the Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) system with the Cas9 nuclease bound to the gRNA and the DNA template to make the genetic alteration.
3 Heat Shock
Heat shock is an essential part of genetic alteration, being responsible for the entry of plasmids. The addition of CaCl₂ neutralizes the phospholipid membrane, facilitating the passage of the plasmid, and Polyethylene glycol 8000 has the characteristic of being able to open pores in the membranes.
When heat shock occurs, the substance is placed at 4ºC for 30 minutes and then placed for 30 seconds in water at 42ºC, with the time and temperature potentially varying according to the experiment. The membrane generates more pores and openings, facilitating the entry of plasmids. For membrane recovery, it is necessary to place it in an incubator at 30ºC with agar to repair the damage and multiply with the new genetic alteration. Bacteria that naturally have this characteristic are naturally competent cells. However, as described above, an artificial procedure was performed to mimic this competent cell characteristic.
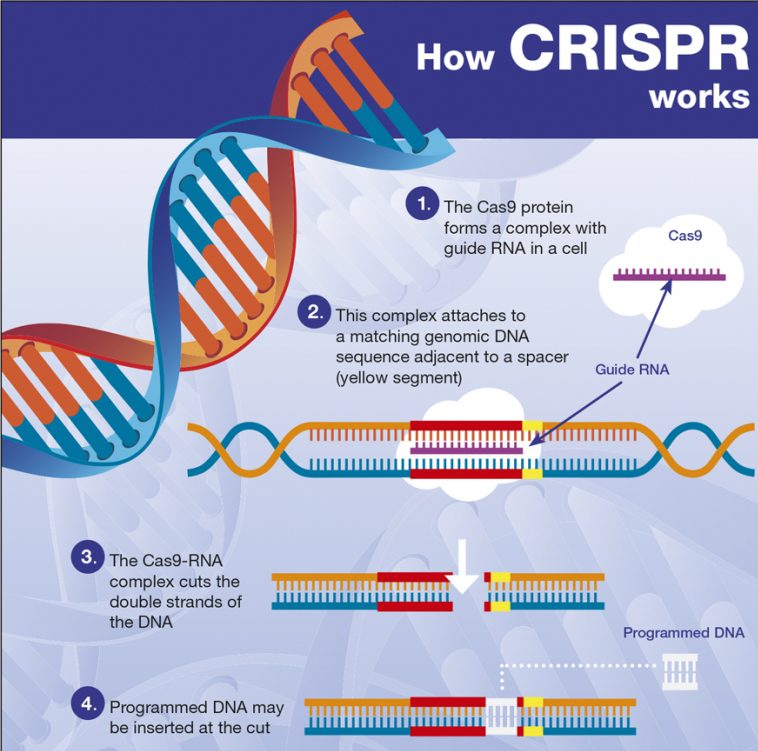
4 CRISPR
Being CRISPR, during evolution, a means of defense for bacteria against viruses from the allocation of a space with the virus gene in a protospacer (PAM), the Cas9 nuclease breaks the nucleotide bond, causing the DNA to repair itself, thus using the RNA that is near it, in this case gRNA along with the DNA template. This mechanism of great evolutionary importance is also used as a gene editing tool.
OBJECTIVES
The main objective of the genetic editing of E. coli bacteria is to highlight the process of genetic alteration, demonstrating the possibility of gene editing using a nuclease and gRNA, using the template of genetic alteration with the guide RNA and the Cas9 nuclease. Being able to create a bacterium resistant to the antibiotic kanamycin, which acts on the ribosomal binding of the bacterium generating incorrect RNA reading, making its growth impossible as it is unable to produce proteins, demonstrating this resistance by cultivating the bacterium in petri dishes with agar and kanamycin.
METHODOLOGY
Based on Karl Popper's hypothetical-deductive method, the hypothesis of genetic alteration using CRISPR that is sought will only be maintained if we manage to falsify the hypothesis. In this case, the verification of the genetic alteration will take place in three tests: E. coli without genetic modification cultivated in agar, E. coli with genetic alteration cultivated in agar with kanamycin, and E. coli without genetic alteration cultivated in agar with kanamycin.
Producing two agar media, one with kanamycin and one without kanamycin, we applied E. coli DH5-Alpha bacteria to both agar media using loops, and then placed both in an incubator at 37ºC. Within 2 days, the bacteria in the medium without kanamycin had reproduced and formed colonies, but the bacteria in the medium with kanamycin had died. We scraped bacteria from the medium without kanamycin with a loop and then carefully vortexed it in a microtube with 100μL of a solution of CaCl₂ and Polyethylene glycol (PEG) 8000, creating competent cells, mixing 10μL of Cas9 plasmids, gRNA, and the DNA template, incubating for 30 minutes at 4ºC and performing a heat shock of 42ºC for 30 seconds, alternating Lysine at position 43 (K43) is transformed into Threonine. The competent cells were added to a microtube of agar with 1.5 ml of water, requiring incubation at 30ºC for 12 hours, after which the competent cells were added to a medium with agar and kanamycin and incubated for another 2 days. We can see in (Fig. 1A) that the bacteria are alive in an agar medium without kanamycin, in (Fig. 1B) that the bacteria with the genetic alteration are alive in an agar medium with kanamycin, and that in (Fig. 1C) the bacteria without genetic alteration are dead in the agar medium with kanamycin.
A.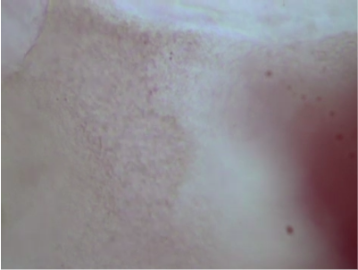 B.
B.
C.
Fig. 1. E. coli cells in agar medium. (A) Live E. coli cells in an agar medium without kanamycin. (B) Genetically altered E. coli cells alive in an agar medium with kanamycin. (C) Dead E. coli cells without being genetically altered and in an agar medium with kanamycin.
More about CRISPR:
Ishino, Y. et al. History of CRISPR-Cas from encounter with a mysterious repeated sequence to genome editing technology. Journal of Bacteriology, 200, 7 (2018).
Jinek M., Chylinski K., Fonfara I., Hauer M., Doudna J.A., Charpentier E. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science. 2012.
